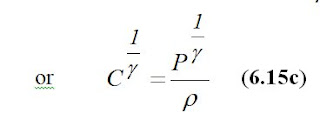pH meters (hydrogen ion concentration)
What we understand under the term of pH? How is it related to the concentration and strength of solutions?
It is known, that acids and bases produce conductive solutions, when dissolve in water, because charged ions are formed. Here are ionisation equations for hydrochloric acid and sodium hydroxide dissolved in water:
Water is formed as the result of this reaction. However, the resulting solution is still conductive, because of the presence of sodium and chloride ions. If the quantities of initial acid and base are equal, then this resultant solution will be neither alkaline, nor acid.
In the case of dissociation of pure water, ions of hydrogen and hydroxyl are formed:
Since the use of ion activities in the form of a power representation is not convenient, a Danish biochemist S.P.L. Sørensen in 1909 proposed to use the expression of pH, which was originally derived from the phrase “power of hydrogen”. The pH is defined as a negative common logarithm (with the base of ten) of the hydrogen ion activity, as follows:
Therefore, the pH of pure water is equal to 7 at 25 °C. Acid solutions contain more hydrogen ions than hydroxyl ions, so the activity of hydrogen ions will be greater than 10-7 , that is, 10-6, 10-5, 10-4, 10-3, etc., with pH equal to 6, 5, 4, 3, etc., respectively. pH values for basic solutions will be 8, 9, 10, 11, etc., respectively.
Instrumentation for pH measurements use an electrometric method. A glass pH-responsive electrode immersed in a solution under measurement will vary electric potential (voltage) on the boundary between the electrode and solution as a function of pH of this solution. However, it is not possible to measure the potential between this electrode and solution only. Why? Because when we connect a measuring device, another potential is developed between the solution and a conductor which connects the measuring device and the solution, this new potential being also dependent on the pH of the solution. Therefore, we need to use one more electrode, the reference electrode, which potential is not dependent on the pH of the solution. In order to make the potential of the reference electrode not dependent on the pH of the solution, it should be filled with a saturated solution.
ES - the overall electromotive force developed in the circuit, mV;
E1 - potential between a silver-silver chloride electrode and the solution 4, mV;
E2 - potential between the solution 4 and an internal surface of the glass electrode, mV;
E3 - potential between mercury and calomel in the reference electrode, mV;
Ex - potential between an outside surface of the glass electrode and the solution under measurement, mV.
R - the gas law constant, R = 8.31451 J/(mole*K);
T - absolute temperature of the solution under measurement, K;
F - Faraday’s number, F = 96485.309 C/mole, C - coulomb.
The overall electromotive force at a constant temperature is the function of the pH of solution only. However, one need to introduce a temperature compensation element (usually a suitable packaged resistor, thermistor, or resistance temperature detector), which is placed close to the glass electrode in the solution under measurement and connected to the electrical circuit for electromotive force measurement.